A. Mean ASDAS and B. mean BASDAI to week 96. Safety set (N = 89).
Por um escritor misterioso
Descrição
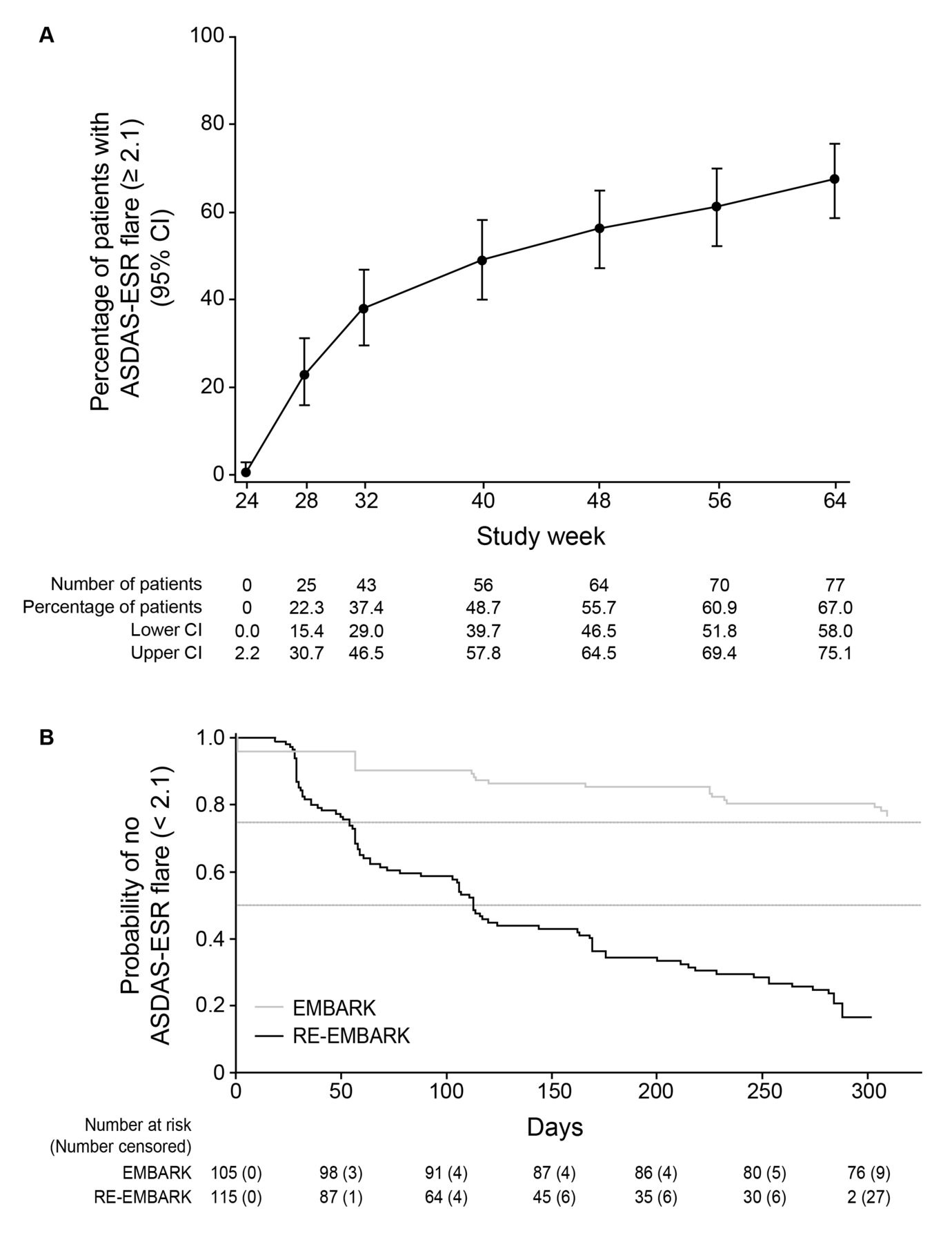
Etanercept Withdrawal and Retreatment in Nonradiographic Axial Spondyloarthritis: Results of RE-EMBARK, an Open-Label Phase IV Trial

Monthly improvements in paid work productivity: ankylosing spondylitis

ASAS-EULAR recommendations for the management of axial spondyloarthritis: 2022 update
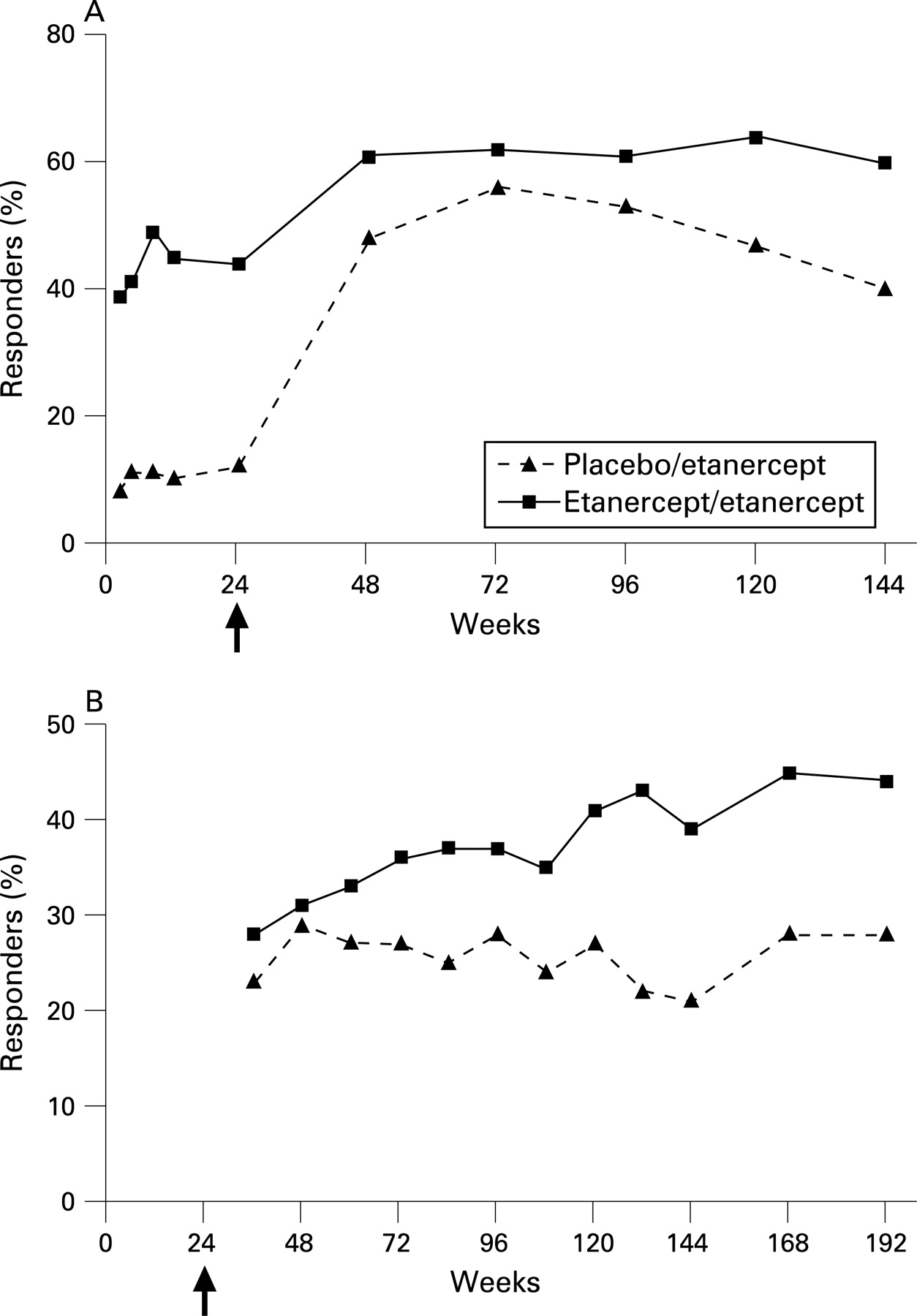
Efficacy and safety of up to 192 weeks of etanercept therapy in patients with ankylosing spondylitis

POS0229 DISEASE ACTIVITY AND INFLAMMATION FOLLOWING WITHDRAWAL OF CERTOLIZUMAB PEGOL TREATMENT IN AXIAL SPONDYLOARTHRITIS PATIENTS WHO DID NOT EXPERIENCE FLARES DURING THE C-OPTIMISE STUDY
Comparative effectiveness of secukinumab and adalimumab in ankylosing spondylitis as assessed by matching-adjusted indirect comparison - Document - Gale OneFile: Health and Medicine

TNF‐alpha inhibitors for ankylosing spondylitis - Maxwell, LJ - 2015

WO2021067465A1 - Treating spondyloarthritic and psoriatic conditions with upadacitinib - Google Patents
A. Mean ASDAS and B. mean BASDAI to week 96. Safety set (N = 89).

These highlights do not include all the information needed to use CYLTEZO safely and effectively. See full prescribing information for CYLTEZO. CYLTEZO® (adalimumab-adbm) injection, for subcutaneous use Initial U.S. Approval: 2017 CYLTEZO (
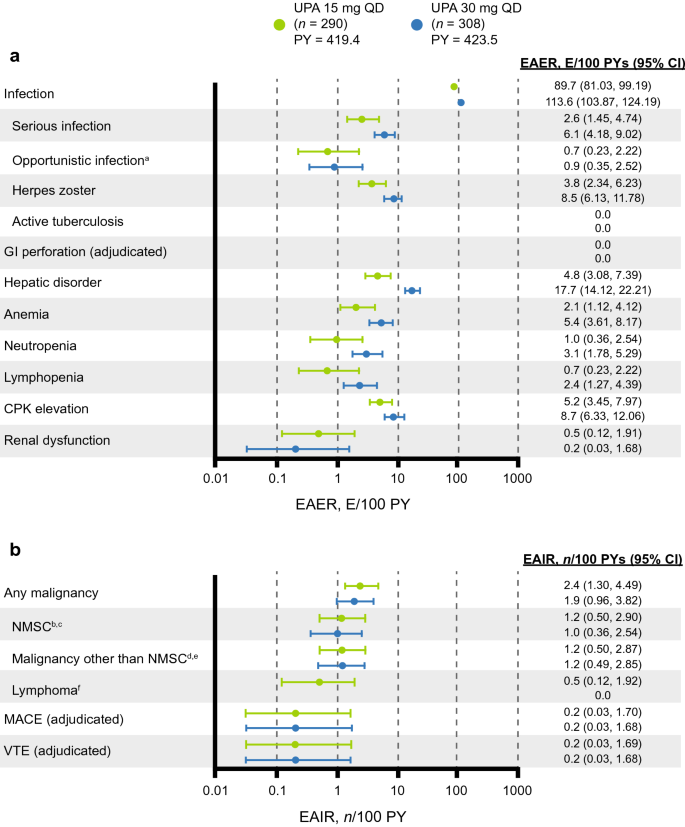
Upadacitinib in Patients with Psoriatic Arthritis and Inadequate Response to Biologics: 56-Week Data from the Randomized Controlled Phase 3 SELECT-PsA 2 Study

THU0379 REDUCTION OF ANTERIOR UVEITIS FLARES IN PATIENTS WITH AXIAL SPONDYLOARTHRITIS FOLLOWING ONE YEAR OF TREATMENT WITH CERTOLIZUMAB PEGOL: 48-WEEK INTERIM RESULTS FROM A 96-WEEK OPEN-LABEL STUDY

TNF Inhibitor Therapy, Ankylosing Spondylitis

Efficacy and safety of continuing versus withdrawing adalimumab therapy in maintaining remission in patients with non-radiographic axial spondyloarthritis (ABILITY-3): a multicentre, randomised, double-blind study - The Lancet
de
por adulto (o preço varia de acordo com o tamanho do grupo)